
On addition of dilute hydrochloric acid, the white precipitate disappeared. (Solved)Ī form four student prepared potassium sulphite solution and divided it into two portions.The first portion gave a white precipitate when reacted with barium nitrate. A form four student prepared potassium sulphite solution and divided it into two portions.The first portion gave a white precipitate when reacted with barium nitrate.Use the electrochemical series below to answer the questions that follow.Ĭalculate the E.M.F for the electrochemical cell represented below. Use the electrochemical series below to answer the questions that follow.(c ) Name one possible solvent that can be used in this experiment / research. (a) Circle the spots which show the banned food colourings. The results showed the presence of banned food colourings in L and M only. The diagram below represents a paper chromatogram for three brands of juices suspected to contain banned food colourings by the Kenya Bureau of Standards. The diagram below represents a paper chromatogram for three brands of juices suspected to contain banned food colourings by the Kenya Bureau of Standards.Calculate the concentration of sulphuric acid in moles per litre.( Na = 23.0 O = 16.0 H = 1.0 ) (Solved)Ģ0.0cm 3 of a solution containing 4gm per litre of sodium hydroxide was neutralized byĨ.0cm 3 of dilute sulphuric acid. Calculate the concentration of sulphuric acid in. 20.0cm 3 of a solution containing 4gm per litre of sodium hydroxide was neutralized byĨ.0cm 3 of dilute sulphuric acid.Using dots (.) and crosses (x), show bonding in:The compound formed when nitrogen reacts with fluorine. Using dots (.) and crosses (x), show bonding in:The compound formed when nitrogen reacts with fluorine.Using dots (.) and crosses (x), show bonding in:-Sodium oxide (Na = 11 O = 8 ) 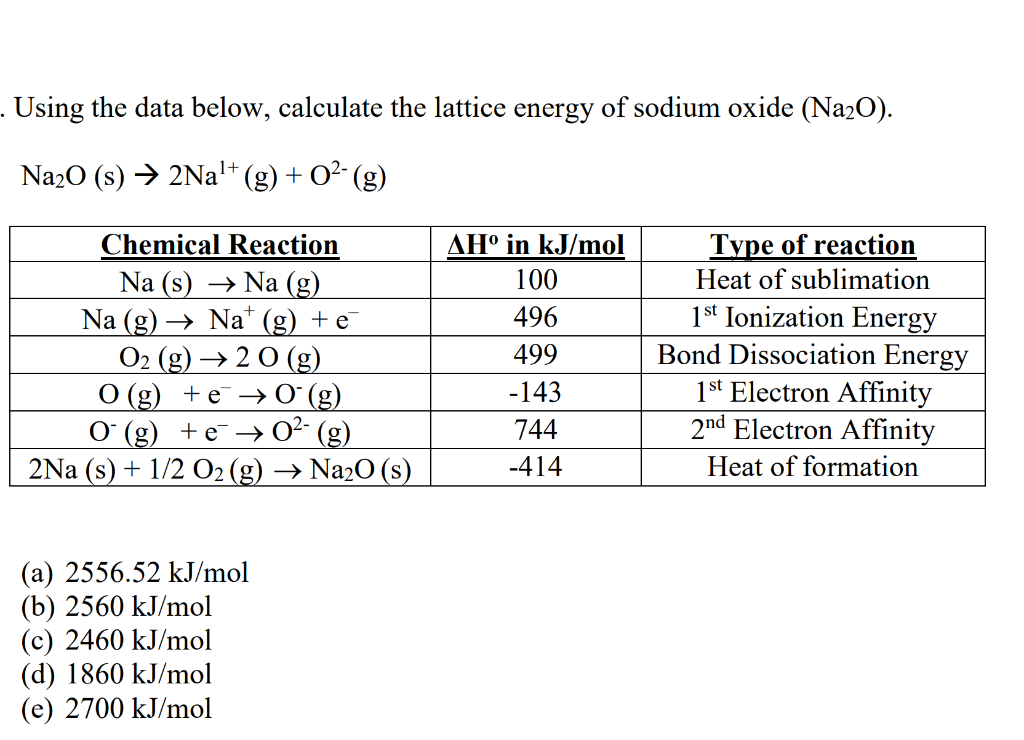
Using dots (.) and crosses (x), show bonding in:-Sodium oxide (Na = 11 O = 8 ) (Solved).What is meant by molar heat of solution ? What is meant by molar heat of solution ? (Solved).If you get a different value, please let me know. Answerĭiscussion: This number has not been checked. 
Evaluate the energy of crystallization, E cryst. Skill: Evaluate the lattice energy and know what values are needed.Īssume the interionic distance for NaCl 2 to be the same as those of NaCl ( r = 282 pm), and assume the structure to be of the fluorite type ( M = 2.512). AnswerĮnergy of crystallization is -3527 kJ/mol Evaluate the energy of crystallization, E cryst for CaO. Lime, CaO, is know to have the same structure as NaCl and the edge length of the unit cell for CaO is 481 pm. Which one of the following has the largest lattice energy? LiCl, NaCl, CaCl 2, Al 2O 3 AnswerĬorrundum Al 2O 3 has some covalent character in the solid as well as the higher charge of the ions. Skill: Explain the trend of lattice energy. Which one of the following has the largest lattice energy? LiF, NaF, CaF 2, AlF 3 Answer When methods to evaluate the energy of crystallization or lattice energy lead to reliable values, these values can be used in the Born-Haber cycle to evaluate other chemical properties, for example the electron affinity, which is really difficult to determine directly by experiment. Much more should be considered in order to evaluate the lattice energy accurately, but the above calculation leads you to a good start.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
